In today’s “Emerging technology startups profiled on SeedSprint” segment, we feature 8 innovative emerging biopharmaceutical companies. These companies are transforming medicine by rethinking their approach to unmet clinical needs and harnessing biological systems to solve them.
With knowledge and tools in biology advancing rapidly, companies are stepping up to apply cutting-edge research to health challenges, old and new. Here are some of the emerging biopharmaceutical companies you’ll find on SeedSprint:
(1) Ambulero (Indication(s): Vascular Disease; Based in Florida, USA): Ambulero is a University of Miami spin-out that is developing novel regenerative treatments for Vascular Disease. Ambulero’s therapies deliver E-selectin to heal damaged vascular tissue via engineered, ‘supercharged MSCs’ or classic AAV vectors. The therapies have the potential to prevent amputation in patients with chronic vascular conditions like Rare Ischemic Disease and Diabetic Critical Limb Ischemia. The company’s cell therapy has received regenerative medicine advanced therapy (RMAT) designation by the FDA. Ambulero is funded by the NIH’s Vascular Interventions/Innovations and Therapeutic Advances (VITA) Program. View SeedSprint profile
(2) Cellestia Biotech (Indication(s): Cancer; Based in Switzerland): Cellestia Biotech is tackling previously untreatable cancers using novel small molecule drugs that target transcription of NOTCH-activated oncogenic genes. The company’s lead candidate, CB-103, inhibits the NOTCH transcription factor complex. CB-103 has a lower toxicity profile than existing treatments like monoclonal antibodies and gamma-secretase inhibitors because it bypasses the gastrointestinal tract. CB-103 is in clinical trials. View SeedSprint profile
(3) NuvOx Pharma (Indication(s): Stroke, Cancer, Covid-19; Based in Arizona, USA): NuvOx Pharma’s NanO2(TM) utilizes the physical properties of dodecafluorpentane to deliver oxygen throughout the lungs, bloodstream, and tissue in microbubbles. In animal studies, NanO2(TM) has shown therapeutic efficacy at a fraction of the dose of other perfluorocarbon delivery methods. NuvOx has secured NIH funding to conduct clinical trials for the treatment of Acute Ischemic Stroke and Glioblastoma. View SeedSprint profile
(4) OrPro Therapeutics (Indication(s): Cystic Fibrosis, Covid-19; Based in California, USA): OrPro’s Theradux® technology treats obstructive airway diseases by targeting thioredoxin to the extracellular surface of epithelial cells in the lung. Thioredoxinan is an anti-inflammatory enzyme with mucous-regulating properties. OrPro’s lead candidate, ORP100S, is an aerosolized formulation of Theradux technology. It can be used with existing home and outpatient devices for easy adoption in patients with Cystic Fibrosis or severe Covid-19. View SeedSprint profile
(5) Vitabolus (Indication(s): Crohn’s Disease, Aging; Based in California, USA): Vitabolus has developed an oral delivery method for cell therapy. The method bypasses the gut to deliver healing cells directly to the intestines. The company’s enteric-coated, acid-resistant capsules have demonstrated efficacy in animal models. Vitabolus’s oral cell therapy is also being investigating for anti-aging, neurological, and inflammatory indications. The company plans to begin clinical studies for Inflammatory Bowel Disease (IBD) soon. View SeedSprint profile
(6) Eurkarys (Indication(s): Cancer, Liver Diseases; Based in France): Eukarys is developing safe, well-tolerated, self-vectorized synthetic gene therapy. The company’s innovative platform technology can be used to treat most human diseases. Eukarys’ first therapy, EUK-LPR, is designed to reduce mortality after partial liver resection by specifically inducing hepatocyte proliferation. The treatment has demonstrated excellent efficacy and tolerance in animals and will enter in regulatory preclinical phase in the next year. The company has two other candidates for the treatment of colorectal cancer and Wilson’s disease. These therapies are currently in the early R&D phase. View SeedSprint profile
(7) GLG Pharma (Indication: Cancer; Based in Massachusetts, USA): GLG Pharma is developing and repurposing small molecule inhibitors to target cancers driven by STAT3 misregulation. The few STAT3 inhibitors currently on the market interfere with signaling molecules, causing many off-target effects and causing safety issues. GLG Pharma’s compounds target the phosphorylation and binding of STAT3 to DNA. As a result, the compounds yield safe toxicity profiles and can even reverse treatment-resistance in cancers. GLG Pharma is currently in clinical trials for Leukemia and Breast Cancer. View SeedSprint profile
(8) Oxidien Pharmaceuticals (Indication(s): Kidney Disease; Based in Florida, USA): Oxidien mitigates kidney stone disease by treating a significant unmet need in hyperoxaluria. The company’s clinically validated enzyme, OX-1, is a dried recombinantly expressed oxalate decarboxylase enzyme that catalyzes the conversion of oxalate to formic acid and carbon dioxide. It is delivered orally with meals to the stomach where OX-1 is highly active and stable in the presence of acid and proteases. The enzyme acts in the gastrointestinal tract but systemically reduces urinary oxalate. The enzyme demonstrated both safety and efficacy in reducing urinary oxalate in healthy volunteers. View SeedSprint profile
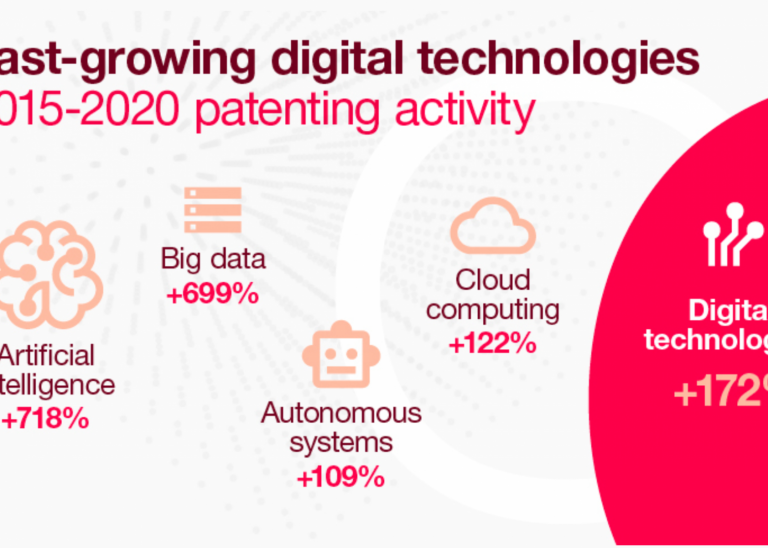
The biopharmaceutical industry has a market size of over $200 billion. With rapid growth expected from the incorporation of tools like artificial intelligence, regenerative therapies, and genetics, the field just keeps getting more exciting.
On SeedSprint, you’ll find over 3,000 biopharmaceutical startup and technology profiles for a diversity of indications. Not on SeedSprint yet? SeedSprint is free of charge for startups and research institutions and available at a flat annual fee for large corporations. You can sign up here!